Conception
- Guyton 11e
- Moore: The Developing Human 8e
Definition
- Synonymous with fertilization; union of sperm and egg to form a zygote
- In the ovary, primary oocyte divides by meiosis and expels a first polar body, thus becoming a secondary oocyte
Generation of primary and secondary oocytes
- In the ovary, primary oocyte divides by meiosis and expels a first polar body, thus becoming a secondary oocyte
- The primary oocyte has 23 pairs of chromosomes and one member of each pair is lost in the polar body
- The secondary oocyte is ovulated into the abdominal cavity and, almost immediately, enters one of the uterine tubes
Intercourse
- Following ejaculation, the semen enters the vagina
- Within 5-10 minutes, the sperms have travelled through the vagina and uterus to the ampulla of the uterine tubes (near the ovarian openings of the tubes); uterine tube = fallopian tube
- Movement of sperms is facilitated by uterine and uterine tube contractions (stimulated by prostaglandins in semen and oxytocin released during female orgasm)
- Nearly half a billion sperm are ejaculated into the vagina, but only a few thousand reach each ampulla
Capacitation of sperm
- Capacitation is the final maturation step that allows sperms to increase motility and prepare for the acrosome reaction; takes place in the female reproductive tract
- Inhibitory factors that are normally present in the male genital ducts are washed away by uterine fluids in the female genital tract
- Once in the uterus, sperms lose membrane cholesterol and the cellular membrane becomes weaker and more permeable to calcium, which is essential for flagellar function. Flagella motility is important for propelling the sperm through the corona radiata and zona pellucida during fertilization.
Fertilization
- Normally takes place in the ampulla of the uterine tubes
- Sperm first penetrates the corona radiata (granulosa cell layers surrounding the ovum), followed by the zona pellucida (glycoprotein membrane around the ovum)
- Acrosome reaction: the acrosome (cap-like structure that overlies the head of the sperm cell) contains large quantities of hyaluronidase and proteolytic enzymes, which help the sperm break through the corona radiata and the zona pellucida
- The acrosome reaction is initiated when the sperm contacts the corona radiata
- Zona reaction: once a sperm penetrates the zona pellucida, the properties of the zona pellucida and plasma membrane change so that it does not allow other sperms to enter
- Acrosome reaction: the acrosome (cap-like structure that overlies the head of the sperm cell) contains large quantities of hyaluronidase and proteolytic enzymes, which help the sperm break through the corona radiata and the zona pellucida
- Once a sperm enters the ovum, the oocyte divides again, expelling a second polar body and forming the mature ovum
- The nucleus of the mature ovum is called the female pronucleus and it contains single copies of 23 chromosomes
- Once the sperm enters the ovum, the head of the sperm swells to form the male pronucleus
- The 23 unpaired chromosomes of the male and female pronuclei align themselves to form a full chromosomal complement in the fertilized ovum
- At the same time (within 30 minutes of the sperm entering the ovum), the two cell membranes fuse to form a single cell called the zygote
Pregnancy
- Williams Obstetrics 23e
- Moore: The Developing Human 8e
Definition
- Intra-uterine development of one or more offspring from a fertilized ovum
Development of the offspring and placenta
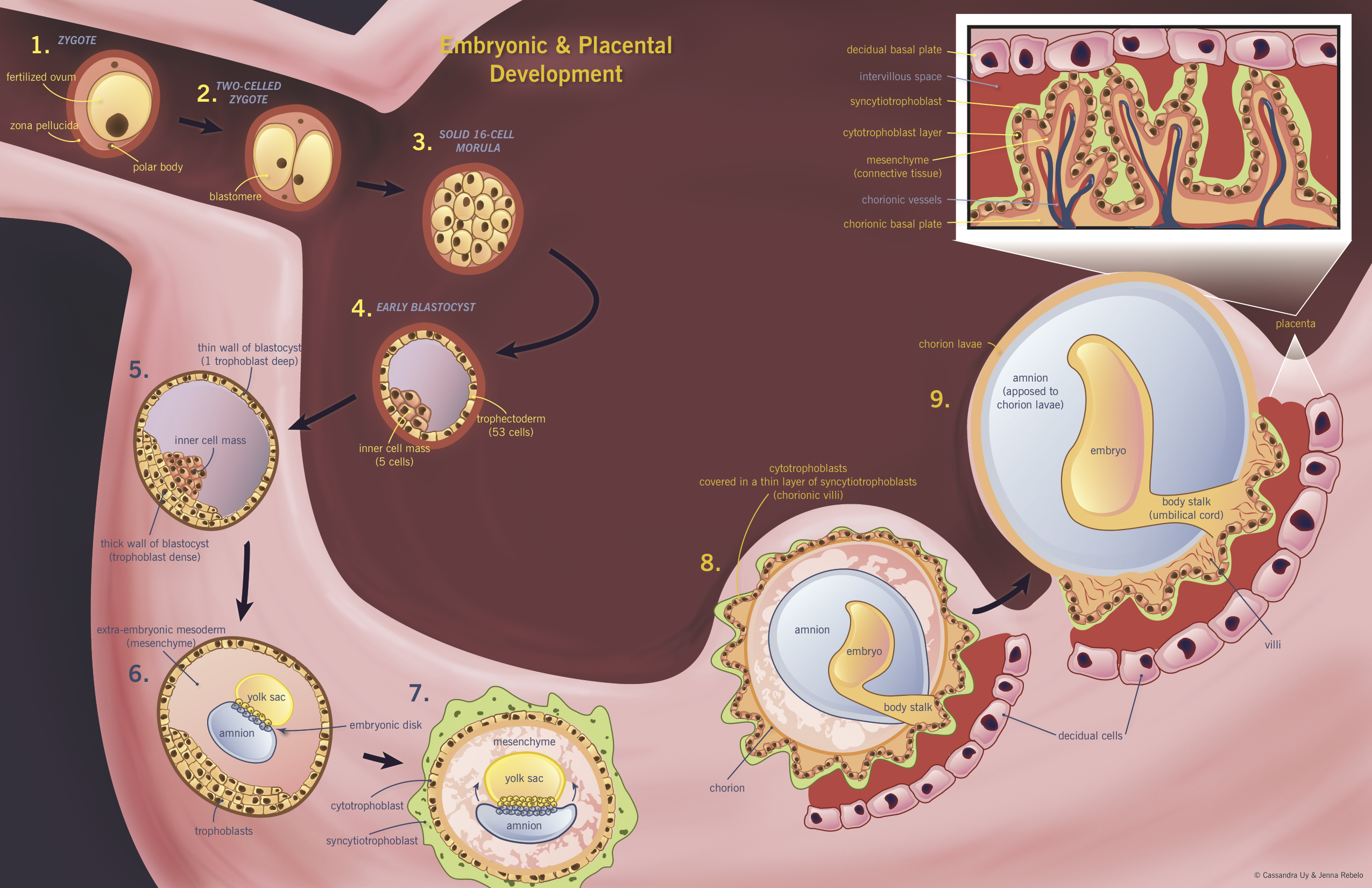
Numbers refer to the diagram below.
- In the uterine tube, the haploid ovum is fertilized and becomes a diploid zygote ①
- Zygote cleaves into two blastomeres (still surrounded by zona pellucida) ②
- Zygote cleavage continues for 3 days
- Blastomere division produces a morula, which is a solid ball of 16 cells ③
- Morula enters the uterine cavity
- Cell division continues and fluid accumulates between the cells of the morula, producing the 58-cell early blastocyst on day 4 post-fertilization ④
- Early blastocyst is composed of an inner cell mass (5 cells that will give rise to embryo) and trophectoderm (53 cells that will give rise to trophoblast)
- After 2 days of free floating in the uterine fluid, the secretory-phase endometrial glands produce proteases that liberate the blastocyst from the zona pellucida (“embryo hatching”)
- Hormones and cytokines produced by the blastocyst now directly influence the endometrium and promote receptivity; the embryo absorbs nutrients from the uterine secretions
- Implantation occurs 6-7 days after fertilization ⑤
- Apposition (phase 1): blastocyst is apposed, in most cases, to the upper posterior uterine wall
- Adhesion (phase 2): increased physical contact between blastocyst and uterine wall
- Invasion (phase 3): blastocyst penetrates the endometrium, upper third of myometrium, and uterine vasculature ⑥
- Whole process is mediated by receptors on the blastocyst and endometrium and by estrogen and progesterone release from the corpus luteum (therefore, uterus is only receptive to implantation between days 20 and 24 of the menstrual cycle)
- By day 8 after fertilization, trophoblast cells have differentiated into the syncytiotrophoblast (multinucleated syncytium that makes up the outer layer of the blastocyst) and the cytotrophoblast (inner layer of mononuclear cells) ⑦
- Trophoblast cells further differentiate into villous and extravillous trophoblast
- Villous trophoblast cells give rise to chorionic villi and are responsible for oxygen and nutrient transport between mother and fetus
- Extravillous trophoblast cells migrate deeper into the uterine wall and penetrate the maternal vasculature (endovascular extravillous cells invade the uterine spiral arteries, while interstitial extravillous cells surround the spiral arteries)
- By day 10, the blastocyst is completely encased in endometrium
- Decidua: endometrial stromal cells which are stimulated by progesterone to swell and store nutrients (the only source of nutrients for the embryo in the first week after implantation)
- As the trophoblast cells are differentiating and invading the endometrium, the inner cell mass differentiates into ectoderm and endoderm
- Shortly thereafter, mesoderm differentiates into mesenchymal cells, which fill the blastocyst cavity
- The embryonic ectoderm gives rise to amnion epithelial cells, which will form the amniotic sac
- The amnion is located on the dorsal surface of the embryo, and the yolk sac on the ventral surface
- The dorsal part of the yolk sac is incorporated into the embryo, forming the gut
- The allantois projects from the yolk sac and later, the hindgut
- As development continues the mesenchymal cells in the blastocyst cavity condense into a body stalk, into which the allantois projects
- The yolk sac is the initial site of hematopoiesis (eventually the liver and bone marrow take over)
- Initially, the amnion covers only the dorsal surface of the embryo, but eventually covers it entirely ⑧
- The amnion has a sub-epithelial layer of mesenchymal cells
- As the amnion grows it obliterates the yolk sac and covers the body stalk
- The amnion eventually fills the intra-uterine space, and the body stalk is unsheathed by the amnion and becomes the umbilical cord
- Meanwhile the syncytiotrophoblast invades the uterine wall
- Interconnecting channels (trophoblastic lacunae) begin to form in the syncytiotrophoblast
- When the decidual capillaries are invaded, the lacunae fill with blood
- The lacunae become larger and merge as the syncytiotrophoblast thins, until the syncytiotrophoblast is just a thin layer overlying the cytotrophoblast cells, and blood fills the space between the syncytiotrophoblast and decidual cells
- One month after fertilization, the uterine spiral arteries are invaded and the blood supply becomes significantly greater
- The cytotrophoblast cells grow into villi that reach towards the decidual cells ⑨
- These are the primary villi, and they grow on all sides of the chorion
- As growth continues, the chorionic villi facing the uterine cavity receive less and less blood, and they degenerate
- The smooth chorion facing the uterine cavity is called the chorion laeve
- As the amnion grows it is apposed to the chorion laeve
- The primary villi are invaded by embryonic mesenchymal cells to form the secondary villi
- The cytotrophoblast cells become less prominent, and the connective tissue stroma more densely packed in order to minimize the distance across the villi
- Finally, the mesenchymal cells undergo angiogenesis, producing chorionic vessels to receive nutrients from the maternal circulation, resulting in tertiary villi
- When the embryonic vessels connect with the chorionic vessels via the umbilical cord, then the true fetal-placental circulation is formed
- In addition to the tertiary villi, there are anchoring villi: extension of syncytiotrophoblast and cytotrophoblasts which connect the chorionic plate to the decidual basal plate (the roof of the intervillous space)
- This is all definitively formed by 8-10 weeks post-fertilization
- Once the embryo fills the uterine cavity, the amnion is opposed to the chorion, which is apposed to the decidua (“the membranes”)
- The amnion provides most of the tensile strength of the membranes
- The amnion lacks smooth muscle cells, nerves, lymphatics, and blood vessels, but is responsible for amniotic fluid homeostasis through salt and water transport
- Initially, the amniotic fluid consists of an ultrafiltrate of maternal plasma, but as the fetus grows fluid diffuses through its skin and begins to influence the makeup of amniotic fluid
- By week 12, fetal kidneys begin to produce urine, which further contributes to the amniotic fluid
- Purpose of the amniotic fluid: it cushions the fetus, allowing movement and musculoskeletal development and minimizing trauma
- The embryonic period begins 2-3 weeks after ovulation, and lasts 8 weeks
- Organogenesis occurs during this period
- At the beginning of the embryonic period, the embryonic disc is well-defined, the body stalk is differentiated, and a true intervillous space exists
- In week 3, fetal blood vessels develop in the chorionic villi
- In week 4, true maternal-fetal circulation and a rudimentary fetal cardiovascular system are established (the heart begins to be partitioned)
- By the end of week 6, the heart is completely formed, fingers and toes are present, and the ears are present as elevations on the head
- The fetal period begins 10 weeks after the LMP and lasts until birth
- Growth and maturation of the organs formed in the embryonic period occur
- Weeks 10-12: fetus can swallow, small intestine can undergo peristalsis and glucose transport
- Week 12: ossification centres appear in bones, genitalia begin to differentiate, skin and nails and hair appear
- Weeks 20-24: neuronal migration and proliferation are completed, and neurologic development shifts towards increasing neuronal organization and differentiation
- Week 24: bronchioles and alveoli are nearly completely developed, but terminal alveolar sacs are typically not yet formed (therefore breathing is unlikely)
- Week 36: deposition of subcutaneous fetal fat occurs
Fetal circulation
- Maternal blood flows through the uterine spiral arteries into the intervillous space of the placenta and is driven by its high pressure up to the chorionic plate
- Invasion of spiral arteries by trophoblasts has resulted in low-resistance vessels that can accommodate the massive increases in blood flow that occur during pregnancy
- Maternal blood bathes the chorionic villi and microvilli, and oxygen and nutrients are transported into the chorionic vessels
- During uterine contractions, blood flow in and out of the placenta is highly limited as the uterine spiral arteries and draining veins are compressed
- The umbilical cord contains two arteries and one vein
- The umbilical vein carries oxygenated blood to the fetus, some of which flows through the fetal liver but most flows through the ductus venosus to the inferior vena cava
- Upon reaching the right atrium, some blood flows through the tricuspid valve to the right ventricle, and some through the foramen ovale into the left atrium and left ventricle
- Blood from the left ventricle flows through the aortic arch and out to the body
- A small amount of blood from the right ventricle flows through the pulmonary arteries and veins, but most flows from the pulmonary artery through the ductus arteriosus to the aortic arch
- Some of the blood flowing through the aortic arch supplies the fetal body, while the remainder flows out through the umbilical arteries to the maternal draining veins
- Many medications and other substances can cross the placenta, and little research has been done on medication safety during pregnancy, so most medications are used off-label
- Certain infections can cross the placenta and infect the fetus
- The TORCH mnemonic is used to remember the most common infections: Toxoplasmosis, Other (coxsackievirus, syphilis, varicella-zoster, HIV, parvovirus B19), Rubella, Cytomegalovirus, Herpes simplex
Maternal physiology
- See table below
Clinical features
- Note that early developmental events are dated from conception, whereas events occurring later in pregnancy are dated from the first day of the last menstrual period
- Small amount of bleeding may occur with implantation, due to invasion of uterine vessels
- Physiologic S3 and supine hypotension may be observed on cardiovascular exam
- By week 12, the uterus should be palpable above the symphysis pubis
- By week 16, gender can be assessed on ultrasound
- After 28 weeks, there is a 90% chance of survival without impairment for a fetus born early without co-morbidities
Definitions
Gestational age (GA): weeks since last menstrual period (LMP)
Estimated date of delivery = LMP + 1 year – 3 months + 7 days (Naegele’s rule)
Term pregnancy: 37-42 weeks GA
- First trimester: 0-12 weeks
- Second trimester: 12-28 weeks
- Third trimester: 28 weeks to delivery
- Preterm: viability to 37 weeks (usually 24-37)
- Postterm: >42 weeks
GTPAL status: abbreviated obstetrical history used for pregnancy risk management
- Gravida (G): # times been pregnant
- Parity: # times given birth, divided into:
- Term (T): # term deliveries
- Preterm (P): # preterm deliveries
- Abortion (A): # spontaneous and therapeutic abortions (
- Living (L): # living
Note: Twin pregnancies count as 1 in each category except for Living.
Maternal physiologic changes
E = estrogen, P = progesterone
| System |
Changes |
Details |
|
Cardiovascular |
Increased cardiac output (CO) CO = HR × SV
|
|
|
Decreased blood pressure (BP) BP = CO × SVR |
|
|
|
Heart is displaced up and to the left |
|
|
|
Respiratory |
Decreased residual volume (RV), functional residual capacity (FRC), and total lung capacity (TLC) |
|
|
Increased resting minute ventilation and tidal volume; unchanged vital capacity |
|
|
|
Decreased PaCO2 |
|
|
|
Renal |
Fluid retention |
|
|
Increased GFR (50%) |
|
|
|
Increased RAAS activity |
|
|
|
Increased renal glucose, amino acid, calcium, and bicarbonate excretion |
|
|
|
Dilation of ureters and renal pelves (R>L) |
|
|
|
Bladder hyperemia and hyperplasia of the muscle and connective tissue |
|
|
|
Blood |
Hemodilution and physiologic anemia |
|
|
Immune depression |
|
|
|
Augmented coagulation and fibrinolysis (in balance) |
|
|
|
Gastrointestinal |
Gastroesophageal reflux disease (GERD) |
|
|
Constipation |
|
|
|
Hemorrhoids |
|
|
|
Cholestasis |
|
|
|
Endocrine |
Anterior pituitary prolactin secretion |
|
|
Thyroid hyperplasia Increased thyroid-binding globulin, total T3 and T4, but free levels unchanged |
|
|
|
Physiological hyperparathyroidism of pregnancy |
|
|
|
Increased serum cortisol |
|
|
|
Peripheral insulin resistance, suppression of glucagon |
|
|
|
Reproductive tract |
Uterus: muscular hypertrophy, increased elastic tissue |
|
|
Cervix: increased vascularity and edema, proliferation of cervical glands and production of mucous plug |
|
|
|
Ovaries: cessation of follicle development and ovulation, relaxin secretion |
|
|
|
Vagina and perineum: hyperemia, connective tissue softening, smooth muscle hypertrophy |
|
|
|
Skin |
Hyperpigmentation on the skin and the linea alba |
|
|
Spider angioma, palmar erythema |
|
|
|
Abdominal striae (striae gravidarum) |
|
|
|
Increased cutaneous blood flow |
|
|
|
Breasts |
Production of colostrum and milk |
|
|
MSK |
Progressive lumbar lordosis |
|
|
Increased mobility of pubic and sacral joints |
|